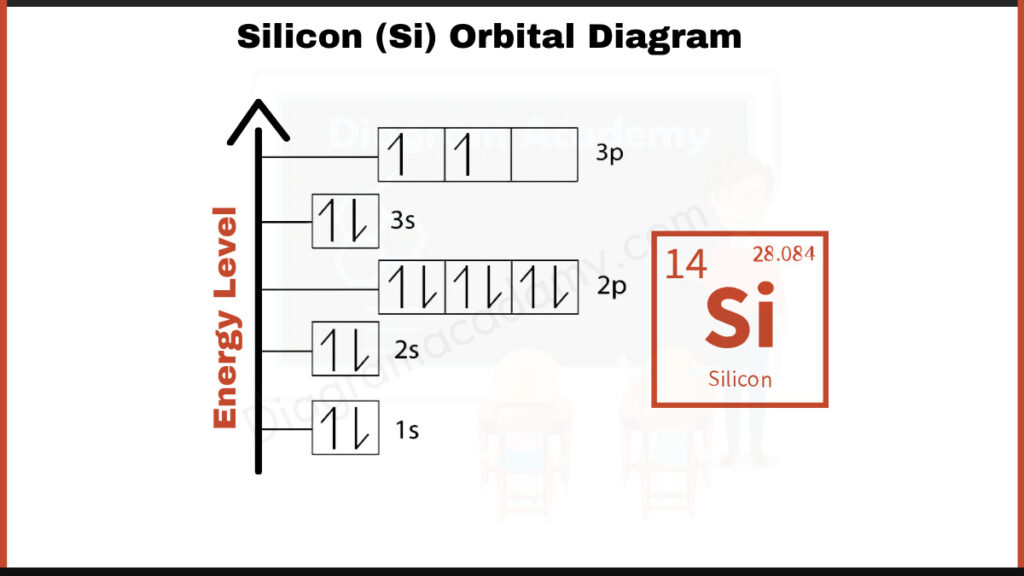
How to write the Orbital Diagram for Silicon?
Silicon (Si) has 14 electrons. They fill shells in a specific order: [2, 8, 4]. This notation represents the number of electrons in each shell. The first shell (1s) holds 2 electrons, the second shell (2s and 2p) holds 8 electrons, and the third shell (3s and 3p) holds the remaining 4 electrons.